CORE MEDICINES PIPELINE
Leading Healthspan Innovations
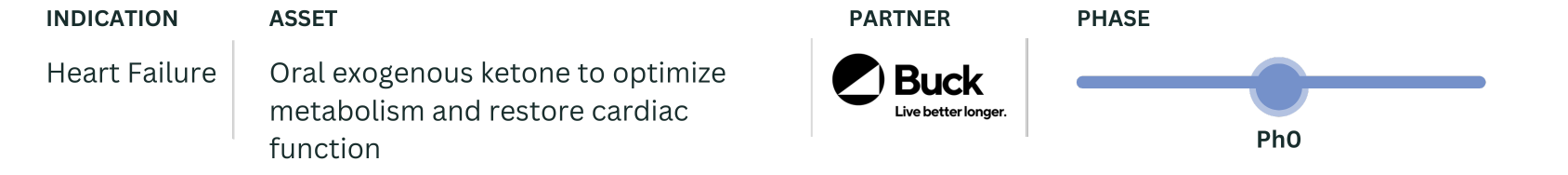
Our core medicines (JuvTherapeutics) are comprised of therapeutics developed by the Juvenescence team, targeting fundamental aging mechanisms with a near-term age-related first indication identified for clinical development.




PORTFOLIO COMPANIES
Our portfolio companies represent strategic investments across a diverse array of technologies focused on increasing healthy lifespan. These span platform technologies, cell therapies & regenerative medicines, as well as nutritional supplement businesses. We work closely with our portfolio companies to enable a cross-pollinating ecosystem, however these companies have their own management teams.
Maximizing Impact Through Diverse Assets
![]()
Foundational platforms based on proprietary Poly-oxazoline (POZ) technology, to deliver small molecules and biologics.
For more info, visit SerinaTherapeutics.com
![]()
Platform tech discovering relationships at the heart of biology to cure disease. Uniquely integrated Lab-in-the-Loop approach combines human genetics, single cell multi-omics directly from human tissue, functional assays & ML to understand the biological processes driving disease. embedding the patient at every level.
For more info, visit RelationRx.com

Utilizing the lymph node as an in-vivo bioreactor to grow functioning ectopic organs. Lead cell therapy is currently in a Ph2a for End-Stage Liver Disease.
For more info, visit LyGenesis.com
![]()
Manipulating the bioelectrome to induce regeneration and treat disease.
For more info, visit Morphoceuticals.com
![]()
Targeted restoration of endogenous metabolites in nutritional products to optimize human health and performance.
For more info, visit www.qitone.com

Synthetic biology company pioneering healthy nutritional interventions.
For more info, visit ChryseaLabs.com
Proprietary digital health platform