08.03.2021
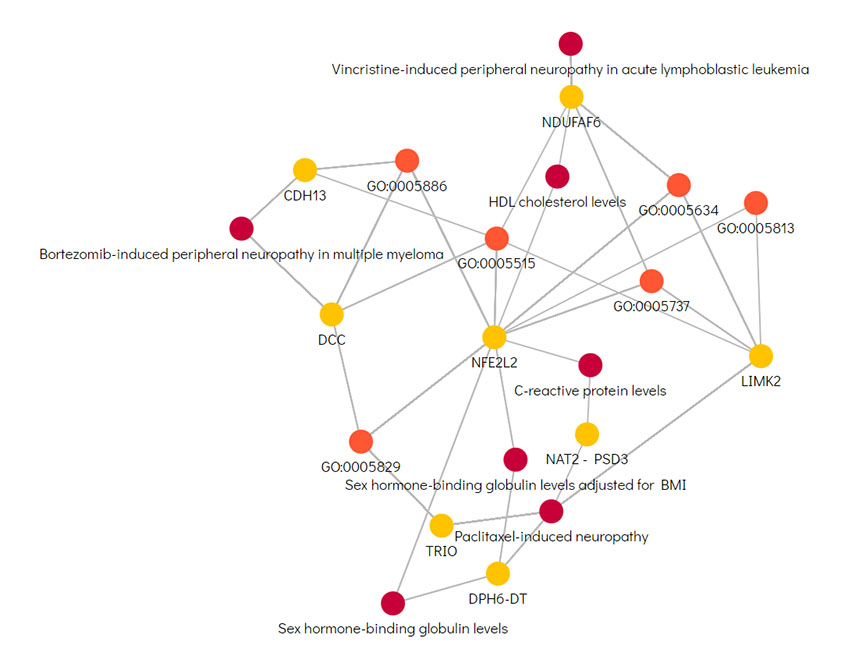
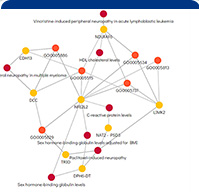
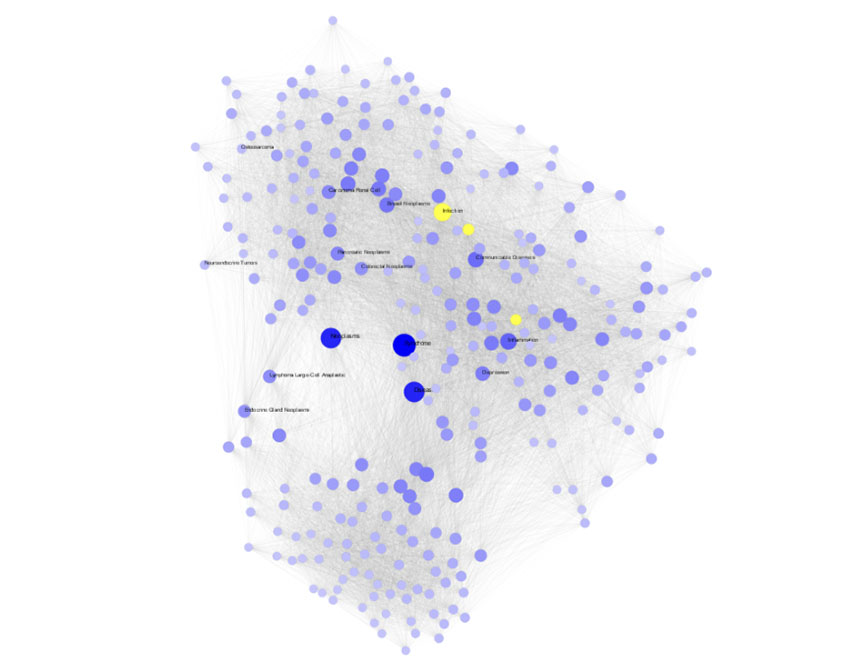
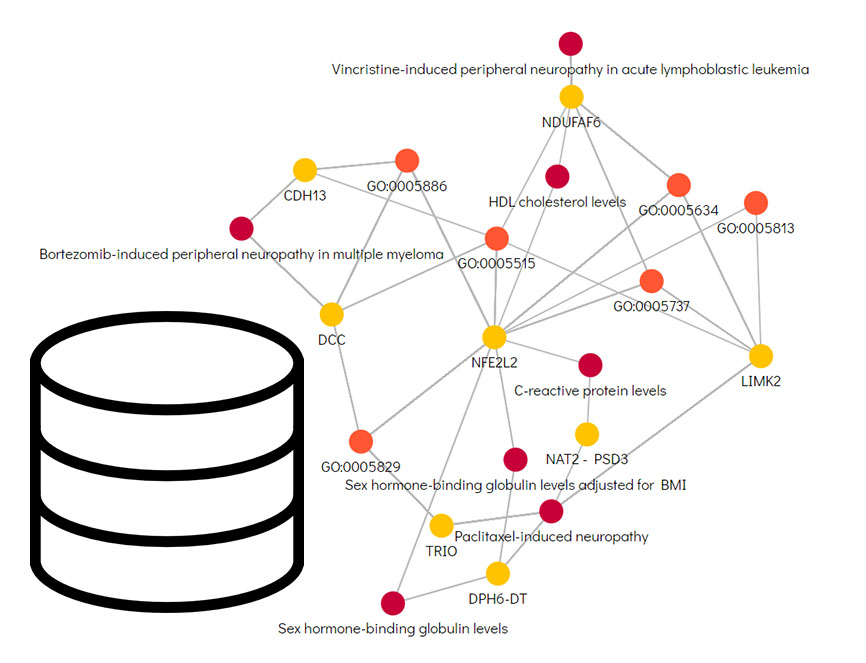
Data Platform For Automated Pharmaceutical Research Using Knowledge Graph
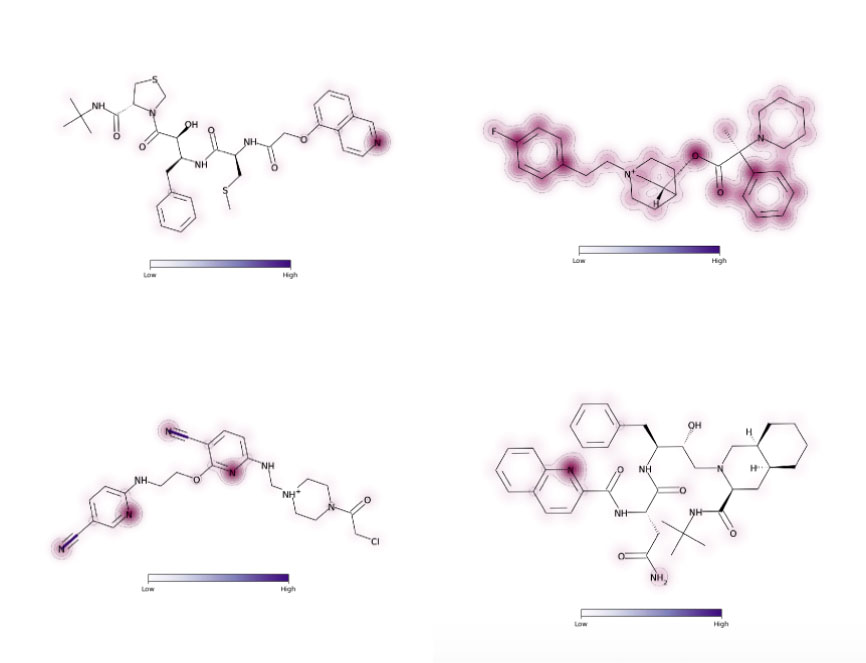
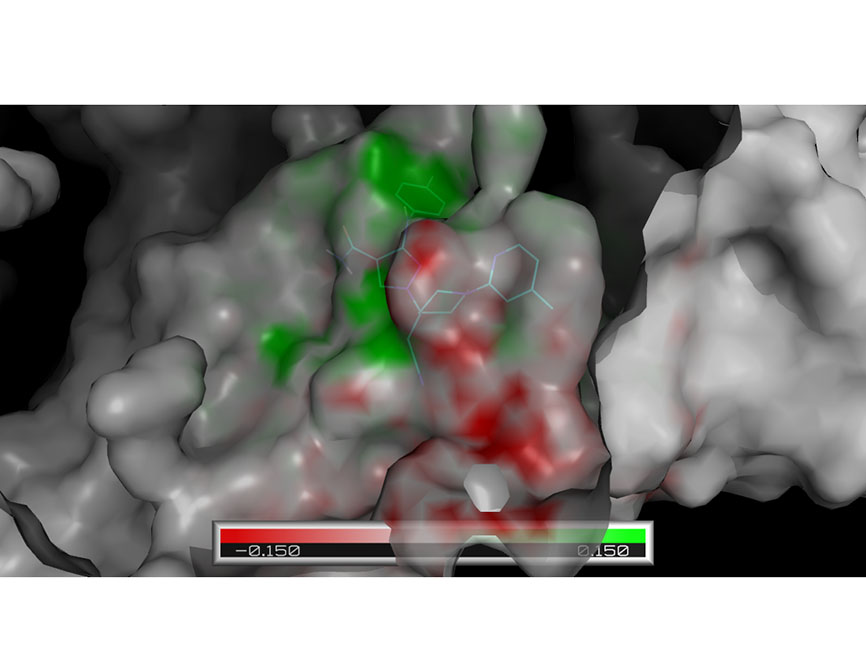
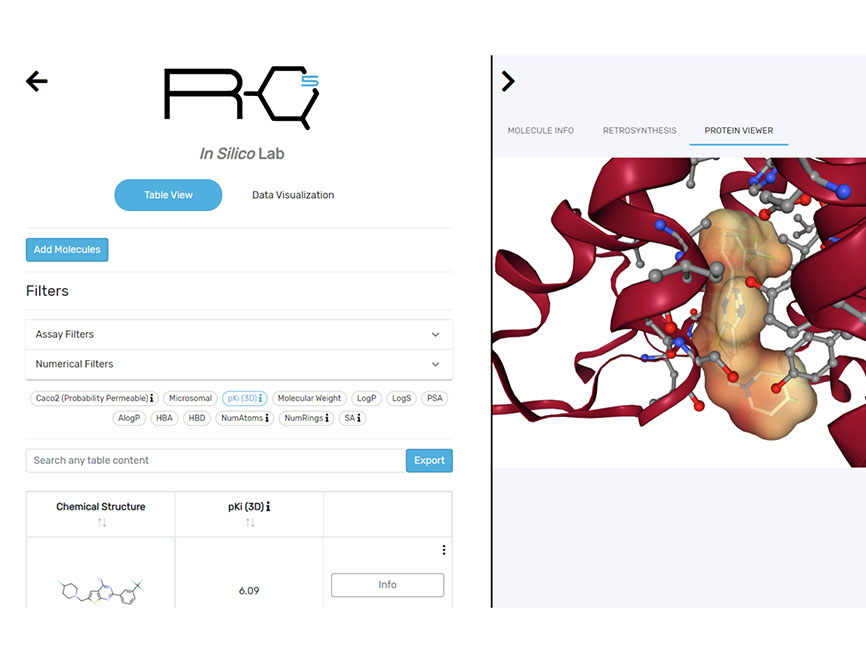
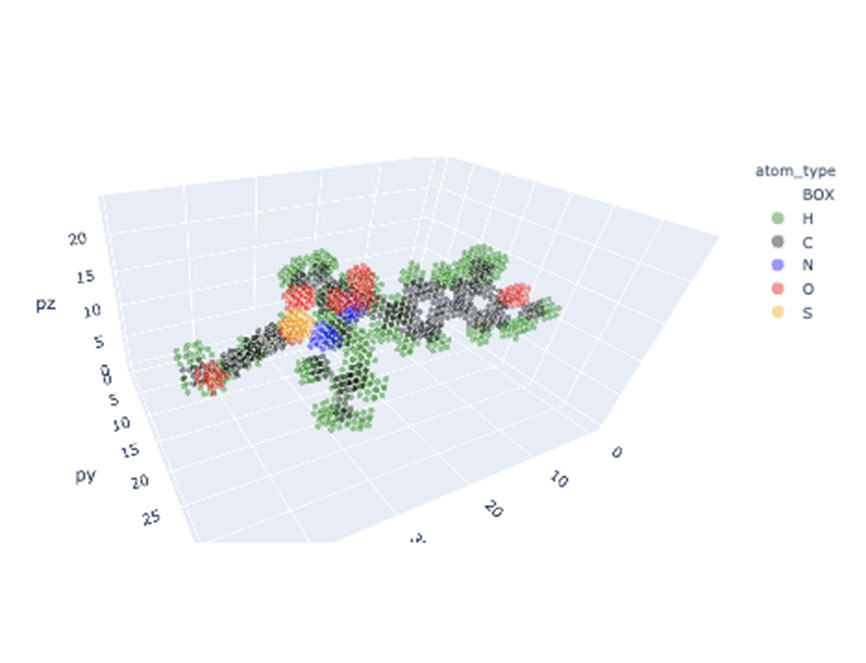
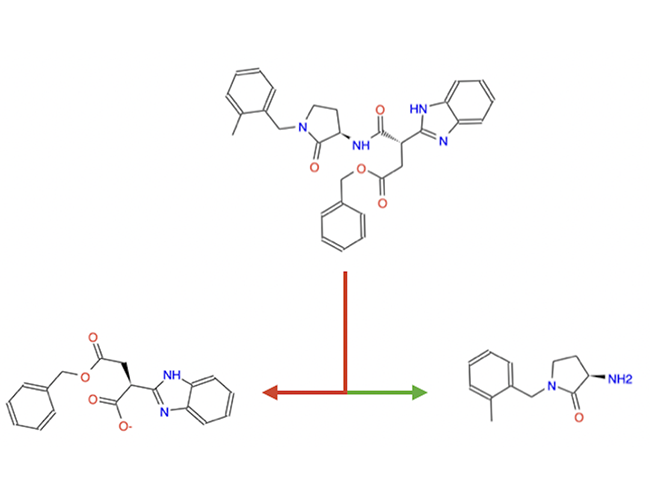
Our computational drug discovery capabilities are enhanced by incorporating state-of-the-art machine learning models and cheminformatics tools to facilitate rapid and efficient discovery and design of novel compounds from hit identification through to lead optimization.